Today, Waggener Edstrom Worldwide is releasing a white paper on digital healthcare titled, “Healthcare Communications in the Digital World: Mitigating the Risks in a Highly Regulated Environment.”
In conjunction with the paper, I will be moderating a live panel discussion this morning, Tuesday, February 24 at 9 AM EST at The Madison, 1177 15th Streeet, NW in Washington, DC. The event will also be available via live webcast.
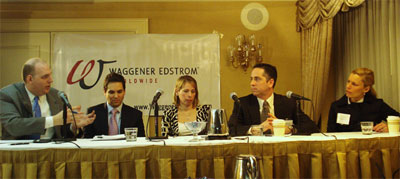
The panel will focus on the online regulatory issues currently facing the healthcare industry, as well as the advances and the promise of Government 2.0. Participants include:
- Mark Gaydos, Sanofi-Aventis US Regulatory Affairs Marketed Products, Senior Director
- Sanjay J. Koyani, U.S. Food and Drug Administration, Director, FDA Web Communications
- Christina Pearson, former U.S. Department of Health and Human Services, Assistant Secretary for Public Affairs
- Jenny Moede, Waggener Edstrom, Healthcare
- David Almacy, Waggener Edstrom, Studio D (Moderator)
From the release:
Following the unprecedented use of social media throughout President Obama’s campaign, the new administration will bring a fresh perspective to digital regulations for healthcare. Companies will need to become educated on how new regulatory policies will affect their online communications. As the administration and the U.S. Food and Drug Administration (FDA) begin to review existing policy and move to create new guidelines, now is the time for pharmaceutical and biotech companies to learn how to effectively engage their audiences online to stay ahead of the competition.
Those who are watching online and may wish to ask a question may do so via Twitter by using the hashtag: #wehealth. Looking forward to great event!
UPDATE
Watch on demand video of the panel discussion.
Download the PDF of the Waggener Edstrom digital healthcare whitepaper.

Topics about Health, Food and Well being » Archive » Digital Healthcare
[…] Professional SAHM placed an observative post today on Digital HealthcareHere’s a quick excerptChristina Pearson, former U.S. Department of BHealth/B and Human Services, Assistant Secretary for Public Affairs… […]